Tolvaptan Tablets
This product is subject to an FDA-approved Risk Evaluation and Mitigation Strategy (REMS). Visit https://www.tolvaptanadpkdsharedrems.com/ for more information.
*All registered trademarks are the property of their respective owners. These products are intended for U.S. residents only.
TOLVAPTAN TABLETS HELP AND SUPPORT
FOR PATIENTS
This webpage helps to explain how to enroll in the Tolvaptan for ADPKD Shared System Risk Evaluation and Mitigation Strategy (REMS). It also outlines how to take tolvaptan tablets to help slow the progression of your autosomal dominant polycystic kidney disease (ADPKD).

Tolvaptan for Autosomal Dominant Polycystic Kidney Disease (ADPKD) Shared System REMS and Medical Monitoring
Your nephrologist or healthcare provider will closely monitor your liver function while you take tolvaptan. For your safety, you must enroll in the Tolvaptan for ADPKD Shared System REMS and undergo regular blood tests. This helps reduce the risk of developing liver problems during treatment.
How to Participate
Before treatment, work with your healthcare provider to:

Tolvaptan for ADPKD Shared System
REMS together.

tablets delivered from a certified pharmacy
participating in the Tolvaptan for ADPKD
Shared System REMS.
Contact the REMS call center number at +1 (866) 244-9446 to sign up by phone or visit Tolvaptan for ADPKD Shared System REMS at www.TolvaptanADPKDSharedREMS.com to enroll online.
How to Monitor
To reduce your risk of liver problems, your healthcare provider will routinely do blood tests to check your liver:

before you start taking tolvaptan tablets,
at 2 weeks and 4 weeks after you start treatment,
then monthly for 18 months during treatment,
and every 3 months from then on.
Schedule for Liver Health Blood Tests
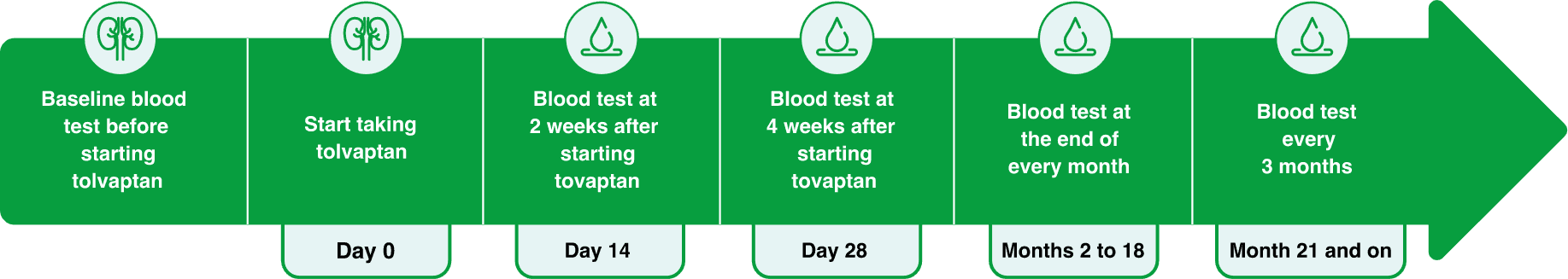
It is important to complete all the required blood testing while on treatment.

While on treatment, watch for any signs of liver problems and inform your healthcare provider if you notice anything unusual:









You can save a guide with key information about the Tolvaptan for ADPKD Shared System REMS, the schedule for blood work,
and the signs or symptoms of liver injury to watch for during treatment.

Tolvaptan Lab Work and Safety Monitoring Guide
Taking Your Tolvaptan Medicine
Tolvaptan tablets are used to slow kidney function decline in adults at risk of rapidly progressing ADPKD.

Your healthcare provider may adjust your tolvaptan dose over time
to help you get the best results from this medicine.
Tablets are taken orally (by mouth) 2 times each day as instructed by your healthcare provider.
Take the first dose when
you wake up and take the
second dose 8 hours later.
Following this schedule ensures
the medicine works throughout
the day and reduces the need
to wake up at night to urinate.
Take your medicine with or
without food.
Be sure to drink enough water
throughout the day so that you
will not get thirsty or become
dehydrated.
Do not eat grapefruit or drink
grapefruit juice while on
tolvaptan, as this could
cause you to have too much
tolvaptan in your blood.
If you miss a dose of tolvaptan tablets, take the next dose at your usual time (do not double up on doses).
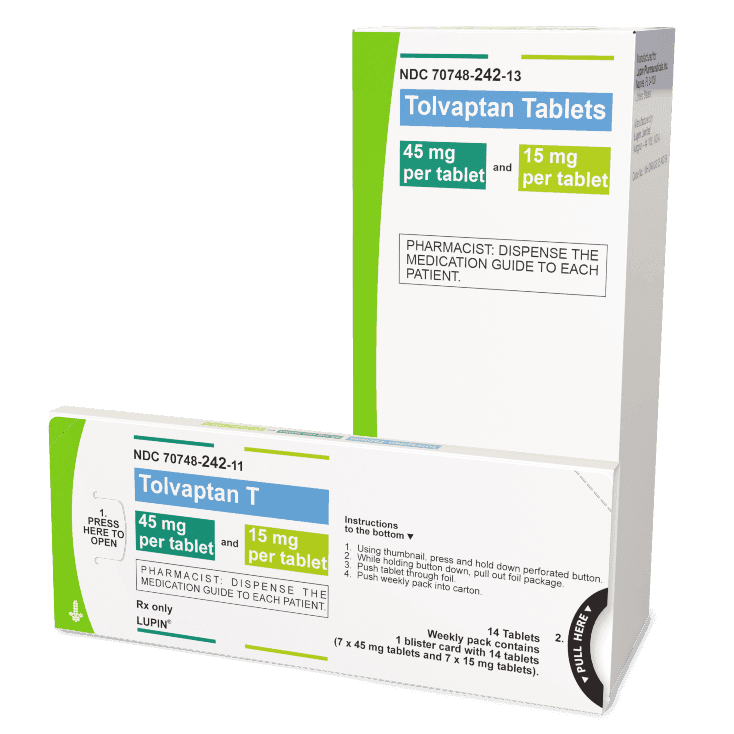
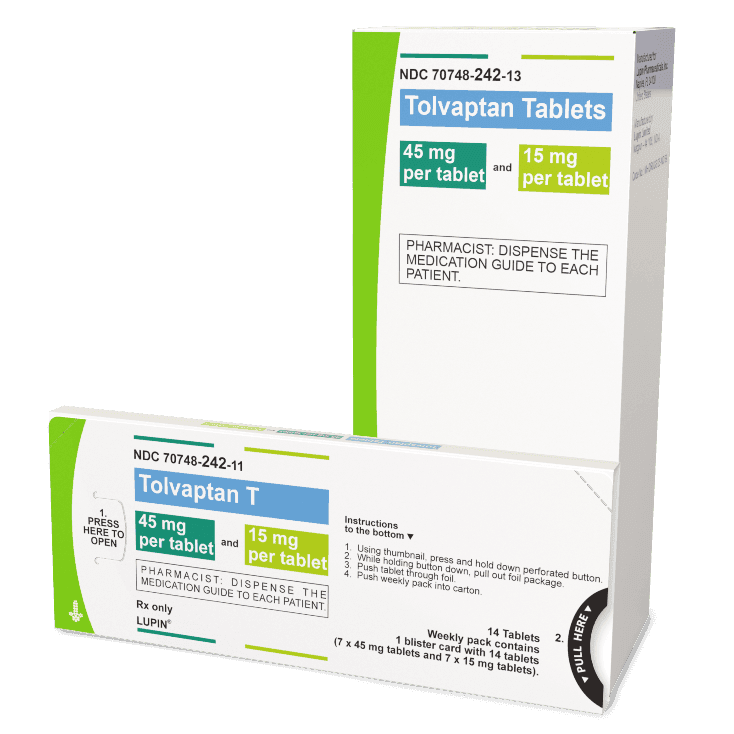
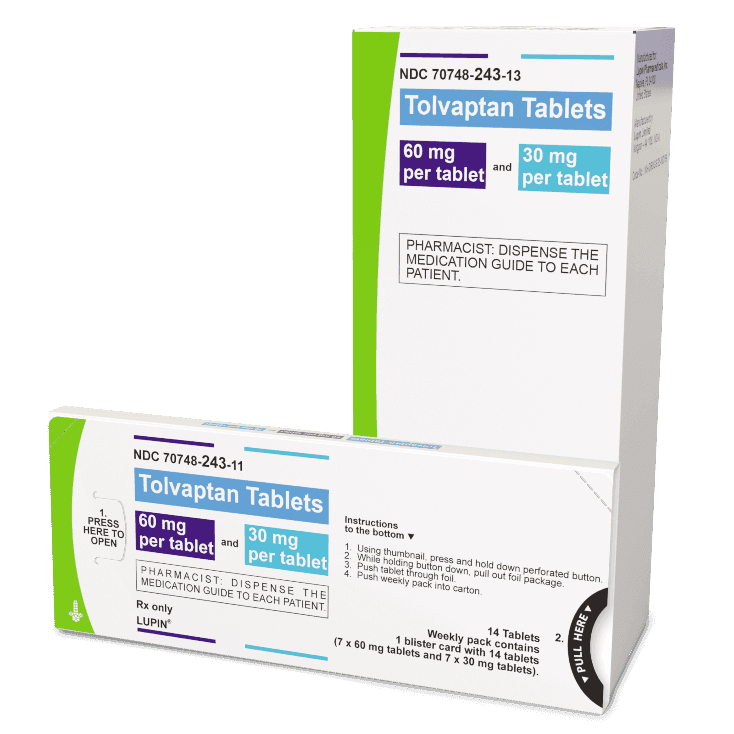
There are 3 dose combinations of tolvaptan tablets available that are provided as weekly blister packs, with the morning and afternoon doses clearly marked.
Starting Dose
(60 mg)


Middle Dose
(90 mg)


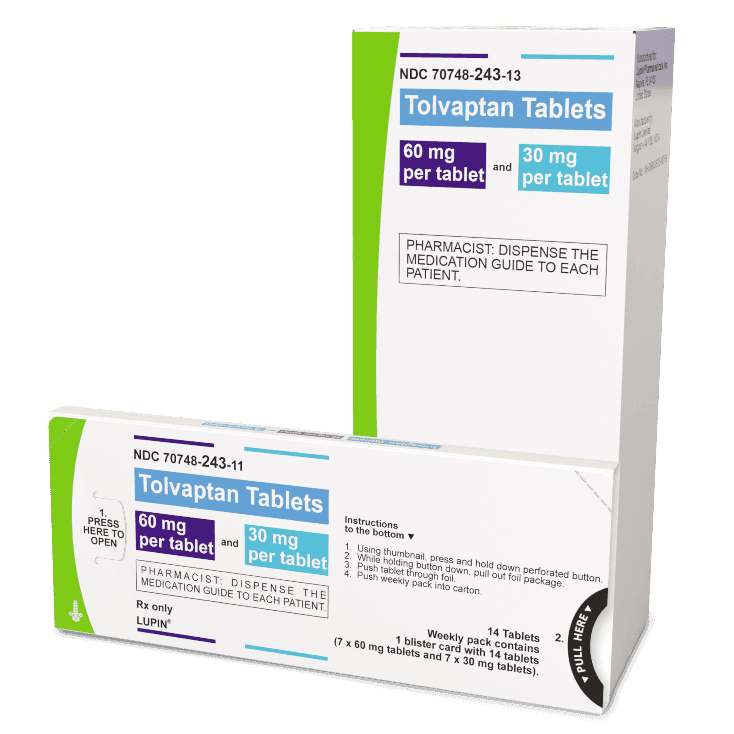
Highest Dose
(120 mg)



Before taking tolvaptan tablets, inform your healthcare provider about all your medical conditions, including:
If you have a history of low sodium levels.
If you are pregnant or plan to become pregnant.
It is not known if tolvaptan will harm your unborn baby. Tell your healthcare provider if you become pregnant or think that you may be pregnant.
If you are breastfeeding or plan to breastfeed.
It is not known if tolvaptan passes into your breast milk. Do not breastfeed during treatment with tolvaptan tablets. Talk to your healthcare provider about the best way to feed your baby during this time.
Also, tell your healthcare provider about all medicines you take, including prescription drugs, over-the-counter medicines, vitamins, and herbal supplements.
Taking tolvaptan tablets with certain medicines could cause you to have too much tolvaptan in your blood.
Tolvaptan tablets should not be taken with certain medications. Your healthcare provider can tell you if it is safe to take tolvaptan tablets with other medicines.
Do not start taking a new medicine without talking to your healthcare provider.
Keep a list of your medicines to show your healthcare provider and pharmacist.
Who Should Not Take Tolvaptan
Do not take tolvaptan tablets if you:
Have a history of liver problems or have signs or symptoms of liver problems, excluding polycystic liver disease
Cannot feel if you are thirsty or cannot replace fluids by drinking
Have been told that the amount of sodium (salt) in your blood is too high or too low
Are dehydrated
Are allergic to tolvaptan or any of the ingredients in tolvaptan tablets
Are unable to urinate


Medication Guide
The Medication Guide includes important questions and answers about
taking tolvaptan tablets to support safe and effective use of this medication.
Tips for Taking Tolvaptan Tablets
To assist you during your treatment, here are some quick tips for taking tolvaptan tablets:

DO
- Take the medicine twice daily (upon waking up and 8 hours later), with or without food,
exactly as instructed by your healthcare provider. - Skip any missed dose and take the next dose at your regular time.
- Store the tablets between 68°F to 77°F and keep them out of reach of children.
- Drink enough water to prevent dehydration.
- Schedule recurring alarms to take each daily dose of medicine on time and set calendar reminders for upcoming blood work.
- Tell your healthcare provider about any changes in your health status.
- Call your healthcare provider or go to the nearest hospital emergency room right away if you take too many tolvaptan tablets.
- Keep a list of your medicines to show your healthcare provider and pharmacist.

DO NOT
- Double up for missed doses.
- Eat grapefruit or drink grapefruit juice during treatment with tolvaptan.
- Start any new medicines, including prescription medicines, over-the-counter medicines, vitamins, and herbal supplements, without speaking to your healthcare provider first.
- Hesitate to ask your healthcare provider any questions about your treatment or health. ADPKD can be hard on your mental health, but you are not in this alone!


Taking Your
Tolvaptan Guide
The Taking Your Tolvaptan Guide provides these helpful
tips about how to take your tolvaptan tablets.
Specialty Pharmacies
Tolvaptan tablets are available to patients by prescription through 5 specialty pharmacies:




Visit these pharmacy sites online or contact these pharmacies directly by phone for any questions about enrollment or your medicine.
For additional savings while on treatment,
ask your Specialty Pharmacy about copay
assistance to lower your out-of-pocket costs.
Frequently Asked Questions
What is the most important information I should know about tolvaptan tablets?
Tolvaptan tablets may cause serious side effects; it’s important to be aware of potential tolvaptan tablet side effects before starting the medication.
Serious liver problems. Tolvaptan tablets can cause serious liver problems that can lead to the need for a liver transplant or can lead to death. Stop taking tolvaptan tablets and call your healthcare provider right away if you get any of the following symptoms:
feeling tired
fever
loss of appetite
rash
nausea
itching
right upper stomach (abdomen) pain or tenderness
yellowing of the skin and white part of the eye (jaundice)
vomiting
dark urine
To help reduce your risk of liver problems, your healthcare provider will do a blood test to check your liver:
before you start taking tolvaptan tablets
at 2 weeks and 4 weeks after you start treatment with tolvaptan tablets
then monthly for 18 months during treatment with tolvaptan tablets
and every 3 months from then on
It is important to stay under the care of your healthcare provider during treatment with tolvaptan tablets.
Because of the risk of serious liver problems tolvaptan tablets are only available through a restricted distribution program called the Tolvaptan Risk Evaluation and Mitigation Strategy (REMS) Program.
Before you start treatment with tolvaptan tablets, you must enroll in the Tolvaptan for ADPKD Shared System REMS, Talk to your healthcare provider about how to enroll in the program.
Tolvaptan tablets can only be dispensed by a certified pharmacy that participates in the Tolvaptan for ADPKD Shared System REMS. Your healthcare provider can give you information on how to find a certified pharmacy
How do I enroll in the Tolvaptan for ADPKD Shared System REMS?
Work with your healthcare provider to sign up. Visit Tolvaptan for ADPKD Shared System REMS for details on how to enroll. Contact the REMS call center directly at +1 (866) 244-9446 for additional information.
How should I take tolvaptan tablets?
Take tolvaptan tablets exactly as your healthcare provider tells you to.
Take tolvaptan tablets orally two times each day.
Take the first dose of tolvaptan tablets when you wake up and take the second dose 8 hours later.
Be sure to drink enough water so that you will not get thirsty or become dehydrated.
If you miss a dose of tolvaptan tablets, take the next dose at your regular time.
If you take too much tolvaptan tablets, call your healthcare provider or go to the nearest hospital emergency room right away.
How should I store tolvaptan tablets?
Tolvaptan tablets come in a child-resistant package. Store tolvaptan tablets between 68°F to 77°F (20°C to 25°C).
Keep tolvaptan tablets and all medicines out of the reach of children.
What are the most serious side effects of tolvaptan tablets?
Tolvaptan tablets may cause serious side effects, including:
Too much sodium in your blood (hypernatremia) and loss of too much body fluid (dehydration). In some cases, dehydration can lead to an extreme loss of body fluid called hypovolemia. You should drink water when you are thirsty and throughout the day and night.
Stop taking tolvaptan tablets and call your healthcare provider if you cannot drink enough water for any reason, such as not having access to water, or vomiting or diarrhea. Tell your healthcare provider if you get any of the following symptoms:
dizziness
fainting
weight loss
a change in the way your heart beats
feeling confused or weak
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
What are the most common side effects of tolvaptan tablets?
The most common side effects of tolvaptan tablets include:
thirst and drinking more fluid than normal
making large amounts of urine, urinating often, and urinating at night
These are not all the possible side effects of tolvaptan tablets.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How do I report a side effect or complaint?
If you have any questions about your health or medicines, talk to your healthcare professional.
To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc. at 1-800-399-2561 or FDA at 1-800-FDA-1088 (www.fda.gov/medwatch).
IMPORTANT SAFETY INFORMATION and INDICATION for TOLVAPTAN TABLET
Serious liver problems. Tolvaptan tablets can cause serious liver problems that can lead to the need for a liver transplant or can lead to death. Stop taking tolvaptan tablets and call your healthcare provider right away if you get any of the following symptoms:
feeling tired
loss of appetite
nausea
right upper stomach (abdomen) pain or tenderness
vomiting
fever
rash
itching
yellowing of the skin and white part of the eye (jaundice)
dark urine
It is important that you tell your healthcare providers that you have received CARVYKTI® and to show them your CARVYKTI® Patient Wallet Card. Your healthcare providers may give you other medicines to treat your side effects.
It is important that you have a blood test before you start tolvaptan tablets to help reduce your risk of liver problems. Your healthcare provider will do a blood test to check your liver:
before you start taking tolvaptan tablets
at 2 weeks and 4 weeks after you start treatment with tolvaptan tablets
then monthly for 18 months during treatment with tolvaptan tablets
and every 3 months from then on
Because of the risk of serious liver problems, tolvaptan tablets are only available through a restricted distribution program called the Tolvaptan Risk Evaluation and Mitigation Strategy (REMS) Program. You must enroll in the REMS program to receive treatment with Tolvaptan.
Do not take tolvaptan tablets if you:
have a history of liver problems or have signs or symptoms of liver problems, excluding polycystic liver disease
cannot feel if you are thirsty or cannot replace fluids by drinking
have been told that the amount of sodium (salt) in your blood is too high or too low
are dehydrated
are allergic to tolvaptan or any of the ingredients in tolvaptan tablets
are unable to urinate
Tell your healthcare provider about all your medical conditions, including if you:
have a history of sodium (salt) levels that are too low or too high.
are pregnant or plan to become pregnant. It is not known if tolvaptan will harm your unborn baby. Tell your healthcare provider if you become pregnant or think that you may be pregnant
are breastfeeding or plan to breastfeed. It is not known if tolvaptan passes into your breast milk. Do not breastfeed during your treatment with tolvaptan tablets. Talk to your healthcare provider about the best way to feed your baby during this time
Tell your healthcare provider about all the medicines you take, including prescription medicines, over-the-counter medicines, vitamins, and herbal supplements.
Taking tolvaptan tablets with certain medicines could cause you to have too much tolvaptan in your blood. Tolvaptan tablets should not be taken with certain medications. Your healthcare provider can tell you if it is safe to take tolvaptan tablets with other medicines
Do not start taking a new medicine without talking to your healthcare provider
Tolvaptan tablets may cause serious side effects, including:
Serious liver problems (see above).
Too much sodium in your blood (hypernatremia) and loss of too much body fluid (dehydration). In some cases, dehydration can lead to extreme loss of body fluid called hypovolemia. You should drink water when you are thirsty and throughout the day and night. Stop taking tolvaptan tablets and call your healthcare provider if you cannot drink enough water for any reason, such as not having access to water, or vomiting or diarrhea. Tell your healthcare provider if you get any of the following symptoms:
dizziness
fainting
weight loss
a change in the way your heart beats
feeling confused or weak
What should you avoid while taking Tolvaptan Tablets?
Do not drink grapefruit juice during treatment with tolvaptan tablets. This could cause you to have too much tolvaptan in your blood.
The most common side effects of tolvaptan tablets are:
thirst and increased fluid intake
making large amounts of urine, urinating often, and urinating at night
These are not all the possible side effects of tolvaptan tablets. Talk to your healthcare provider about any side effect that bothers you or that does not go away. For more information, ask your healthcare provider or pharmacist.
If you have any questions about your health or medicines, talk to your healthcare professional.
To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc. at 1-800-399-2561 or FDA at 1-800-FDA-1088 (www.fda.gov/medwatch).
INDICATION:
What is Tolvaptan Tablet?
Tolvaptan Tablet is a prescription medicine used to slow kidney function decline in adults who are at risk for rapidly progressing autosomal dominant polycystic kidney disease (ADPKD). It is not known if Tolvaptan Tablet is safe and effective in children.
Please read FULL PRESCRIBING INFORMATION, including BOXED WARNING, and MEDICATION GUIDE.
PP-TOL-0001
©2025 Lupin Pharmaceuticals, Inc.
TOLVAPTAN TABLETS HELP AND SUPPORT
FOR PROVIDERS
Tolvaptan tablets are the generic equivalent of JYNARQUE® (tolvaptan) tablets. Patients with autosomal dominant polycystic kidney disease (ADPKD) now have access to an autosomal dominant polycystic kidney disease medication that helps slow the decline of kidney function at generic pricing.
Tolvaptan tablets are available only through the Tolvaptan for ADPKD Shared System Risk Evaluation and Mitigation Strategy (REMS) due to the risk of serious liver injury. This Tolvaptan for ADPKD Shared System REMS focuses on patient monitoring to reduce liver injury risks.
This page will help you learn how to enroll in the Tolvaptan for ADPKD Shared System REMS and provide more information about tolvaptan tablets.


To prescribe tolvaptan tablets for your patients with ADPKD, follow these steps to enroll yourself and your patients in the Tolvaptan for ADPKD Shared System REMS before starting treatment.
Perform baseline liver function tests (ALT, AST, Bilirubin).
Counsel patients about the Tolvaptan for ADPKD Shared System REMS and liver injury risks.
Help your patients to enroll in the Tolvaptan for ADPKD Shared System REMS.
Monitor patients for liver injury symptoms and perform liver function tests at:
2 weeks after starting treatment,
4 weeks after starting treatment,
monthly for the first 18 months, and
every 3 months thereafter.

It is important for patients who take tolvaptan tablets to be aware of potential side effects associated with liver injury. Proper management and prompt action can mitigate further complications.
Advise patients to stop taking tolvaptan tablets immediately and inform you if they experience symptoms of liver injury, such as fatigue, anorexia, nausea, upper right abdominal discomfort or tenderness, vomiting, fever, rash, itching, jaundice, or dark urine.
REMS-certified Pharmacies
Tolvaptan tablets are available to patients by prescription through 5 REMS-certified pharmacies:




Contact the REMS call center at +1 (866) 244-9446 for more information or visit Tolvaptan for ADPKD Shared System REMS for details.

Tolvaptan REMS
Reference Guide
Tolvaptan Dosing
Tolvaptan tablets are indicated to slow kidney function decline in adults at risk of rapidly progressing autosomal dominant polycystic kidney disease. Instruct patients to take tolvaptan twice daily: once upon waking and again 8 hours later. Tablets are administered orally, and patients are advised to:
take it with or without food,
drink plenty of water to prevent thirst or dehydration, and
avoid grapefruit and grapefruit juice beverages while taking tolvaptan tablets.
If a dose is missed at the scheduled time, instruct patients to take the next dose at the usual time without doubling up on doses.
Titrating Tolvaptan Doses
Doses are adjusted weekly, if tolerated, up to the target maintenance dose of 120 mg daily. There is a minimum of 1 week between each titration, if tolerated.
Initial Dose
(60 mg)

after waking

8 hours later*
Titration Dose
(90 mg)

after waking

8 hours later*
Target (Maintenance)
Dose (120 mg)

after waking

8 hours later*

*Mitigates the need to wake up to urinate.
Adjust patient doses up or down based on tolerability. Reduce tolvaptan doses for patients using moderate CYP3A inhibitors.
| Standard Dose | Adjusted Dose if Co-administered With Moderate CYP3A Inhibitors | ||
|---|---|---|---|
| Morning | Afternoon | Morning | Afternoon |
| 90 mg | 30 mg | 45 mg | 15 mg |
| 60 mg | 30 mg | 30 mg | 15 mg |
| 45 mg | 15 mg | 15 mg | 15 mg |
Contraindications
Tolvaptan tablets should not be used by patients who:
have a history, signs, or symptoms of significant liver impairment or injury (other than uncomplicated polycystic liver disease),
are taking strong CYP3A inhibitors,
have uncorrected abnormal blood sodium concentrations,
are unable to sense or respond to thirst,
have hypovolemia,
experience hypersensitivity (e.g., anaphylaxis, rash) to tolvaptan or any of its ingredients,
have uncorrected urinary outflow obstruction, or
have anuria.

Guide patients through their treatment so they know what to expect. Use these helpful counseling tips to support patients effectively:
Clarify that tolvaptan slows kidney function decline in adults with rapidly progressing ADPKD.
Instruct patients to take the first tolvaptan dose upon waking in the morning and the second dose 8 hours later in the afternoon.
Help them set alarms or reminders to take doses on time.
Encourage them to drink plenty of water and avoid eating grapefruit or drinking grapefruit juice.
Explain what the Tolvaptan for ADPKD Shared System REMS is and how close monitoring helps keep them safe while on treatment.
Review the monitoring schedule together and suggest that they schedule lab test appointments on their calendars.
Ask patients to tell you about any changes in their health status or medications before making any dose adjustments.
Remind them to contact you with any questions or concerns during treatment.
Living with ADPKD can be emotionally challenging. Let patients know that you are here for them.
Review the Medication Guide with your patients. It provides answers to common questions to help reduce the risk of liver problems.
Ask patients if they have questions about tolvaptan tablets. Provide them with these helpful resources for easy reference:
Tolvaptan Guide
Share this dos and don’ts list summarizing key administration information to help your patients safely take their medication.

Safety Monitoring Guide
Review this guide with your patients to help set expectations for monitoring while on their treatment. This summary introduces the Tolvaptan for ADPKD Shared System REMS, outlines required lab monitoring, and highlights signs or symptoms of liver injury to watch for during treatment.

ADPKD can be challenging. Advocacy and patient support groups can provide emotional support, valuable information, and advice to help your patients along their treatment journey with tolvaptan tablets.
Encourage your patients to reach out to
their REMS-certified Pharmacy directly for
more information about copay assistance.
Frequently Asked Questions
How do I enroll in the Tolvaptan for ADPKD Shared System REMS?
Refer to Tolvaptan for ADPKD Shared System REMS at www.TolvaptanADPKDSharedREMS.com for details on how to enroll or contact the REMS call center directly at +1 (866) 244-9446.
How do I report a side effect or complaint?
You can contact LUPIN to report a side effect or a product complaint at drugsafety@lupin.com or call 1-800-399-2561.
You may also report side effects to the U.S. Food and Drug Administration (FDA) at www.fda.gov/medwatch or call 1-800-FDA-1088.
INDICATION and IMPORTANT SAFETY INFORMATION
Tolvaptan tablets are indicated to slow kidney function decline in adults at risk of rapidly progressing autosomal dominant polycystic kidney disease (ADPKD).
Tolvaptan tablets can cause serious and potentially fatal liver injury. Acute liver failure requiring liver transplantation has been reported
Measure transaminases (ALT, AST) and bilirubin before initiating treatment, at 2 weeks and 4 weeks after initiation, then monthly for the first 18 months and every 3 months thereafter. Prompt action in response to laboratory abnormalities, signs, or symptoms indicative of hepatic injury can mitigate, but not eliminate, the risk of serious hepatotoxicity Because of the risks of serious liver injury tolvaptan tablets are available only through a Risk Evaluation and Mitigation Strategy program called
Because of the risks of serious liver injury tolvaptan tablets are available only through a Risk Evaluation and Mitigation Strategy program called the Tolvaptan for ADPKD Shared System REMS. Prescribers, Pharmacies and patients must enroll in the REMS program.
CONTRAINDICATIONS:
History, signs or symptoms of significant liver impairment or injury. This contraindication does not apply to uncomplicated polycystic liver disease
Taking strong CYP3A inhibitors
With uncorrected abnormal blood sodium concentrations
Unable to sense or respond to thirst
Hypovolemia
Hypersensitivity (e.g., anaphylaxis, rash) to tolvaptan tablets or any component of the product
Uncorrected urinary outflow obstruction
Anuria
Serious Liver Injury: Tolvaptan tablets can cause serious and potentially fatal liver injury. Acute liver failure requiring liver transplantation has been reported in the post-marketing ADPKD experience. Discontinuation in response to laboratory abnormalities or signs or symptoms of liver injury (such as fatigue, anorexia, nausea, right upper abdominal discomfort, vomiting, fever, rash, pruritus, icterus, dark urine or jaundice) can reduce the risk of severe hepatotoxicity. To reduce the risk of significant or irreversible liver injury, assess ALT, AST and bilirubin prior to initiating tolvaptan tablets, at 2 weeks and 4 weeks after initiation, then monthly for 18 months and every 3 months thereafter.
Hypernatremia, Dehydration and Hypovolemia: Tolvaptan tablets therapy increases free water clearance which can lead to dehydration, hypovolemia and hypernatremia. Instruct patients to drink water when thirsty, and throughout the day and night if awake. Monitor for weight loss, tachycardia and hypotension because they may signal dehydration. Ensure abnormalities in sodium concentrations are corrected before initiating therapy. If serum sodium increases above normal or the patient becomes hypovolemic or dehydrated and fluid intake cannot be increased, suspend tolvaptan tablets until serum sodium, hydration status and volume status parameters are within the normal range.
Inhibitors of CYP3A: Concomitant use of tolvaptan tablets with drugs that are moderate or strong CYP3A inhibitors (e.g., ketoconazole, itraconazole, lopinavir/ritonavir, indinavir/ritonavir, ritonavir, and conivaptan) increases tolvaptan exposure. Use with strong CYP3A inhibitors is contraindicated; dose reduction of tolvaptan tablets are recommended for patients taking moderate CYP3A inhibitors. Patients should avoid grapefruit juice beverages while taking tolvaptan tablets.
Adverse Reactions: The most common reasons for discontinuation of Tolvaptan tablets in a randomized trial were polyuria, pollakiuria and nocturia. The most common treatment emergent adverse reactions in the same trial (>3% and >1.50/0 of placebo) were: thirst, dry mouth, fatigue, dizziness, dyspepsia, decrease appetite, abdominal distention, dry skin, hyperuricemia, and palpitations.
Other Drug Interactions:
Strong CYP3A Inducers: Co-administration with strong CYP3A inducers reduces exposure to tolvaptan tablets. Avoid concomitant use of tolvaptan tablets with strong CYP3A inducers
V2-Receptor Agonist: Tolvaptan interferes with the V2-agonist activity of desmopressin (dDAVP). Avoid concomitant use of tolvaptan tablets with a V2-agonist
Pregnancy and Lactation: Based on animal data, tolvaptan tablets may cause fetal harm. In general, tolvaptan tablets should be discontinued during pregnancy. Advise women not to breastfeed during treatment with tolvaptan tablets.
To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc. at 1-800-399-2561 or FDA at 1-800-FDA-1088 (www.fda.gov/medwatch).
Please read FULL PRESCRIBING INFORMATION, including BOXED WARNING, and MEDICATION GUIDE.
PP-TOL-0004
©2025 Lupin Pharmaceuticals, Inc.
Professionals. Please confirm:

Global Privacy Control Detected
We have detected your Global Privacy Control (GPC) signal. In accordance with applicable privacy laws, we have automatically honored your preference and disabled all non-essential cookies and tracking.
Only necessary cookies required for the website to function are enabled.
Opt-Out Request Honored
We have respected your privacy preference. Your opt-out request through cookie settings or Global Privacy Control signal has been honored.






 Download the Tolvaptan Lab Work and Safety Monitoring Guide
Download the Tolvaptan Lab Work and Safety Monitoring Guide




