Mumbai, February 09, 2023: Pharma major Lupin Limited [BSE: 500257 | NSE: LUPIN] reported its financial performance for the quarter ending Dec 31, 2022 today. These unaudited results were taken on record by the Board of Directors at a meeting held today.
| Financial Highlights – Consolidated IND-AS |
Amt in INR mn
| Particulars | Quarter |
| Q3 FY2023 | Q2FY2023 | QoQ Growth % | Q3 FY2022 | YoY Growth % |
| Sales | 42,446 | 40,912 | ↑3.7% | 40,875 | ↑3.8% |
| EBITDA | 5,506 | 4,680 | ↑17.6% | 4,039 | ↑ 36.3% |
| EBITDA Margin (%) | 13.0% | 11.4% | ↑160 bps | 9.9% | ↑ 310 bps |
| PBT | 2,461 | 2,096 | ↑ 17.4% | 1,671 | ↑ 47.3% |
Income Statement highlights – Q3 FY2023
- Gross Profit was INR 25,375 mn compared to INR 23,784 mn in Q2 FY2023, with gross margin at 59.8%
- Personnel cost was 18.0% of sales at INR 7,640 mn compared to INR 7,716 mn in Q2 FY2023
- Manufacturing and other expenses were 31.5% of sales at INR 13,351 mn compared to INR 12,268 mn in Q2 FY2023
- Investment in R&D for the quarter was INR 2,896 mn (6.8% of sales)
Balance Sheet highlights
- Operating working capital was INR 63,837 mn as on Dec 31, 2022
- Capital Expenditure for the quarter was INR 1,555 mn
- Net Debt as on Dec 31, 2022 stands at INR 33,479 mn
- Net Debt-Equity for the company as on Dec 31, 2022 stands at 0.27
Commenting on the results, Mr. Nilesh Gupta, Managing Director, Lupin Limited said, “We continued to build on our momentum in the quarter with growth in sales and improvement in profitability. Sales in the U.S. have improved on the back of new product launches and the brands acquisition we made during the quarter. Excluding the impact of genericization on our diabetes portfolio, the India business has performed in line with the market. With the recent sales force expansion and new product launches, we expect to be back to above-market growth. We are committed to executing on our strategic initiatives to drive long-term growth and profitability, and maintaining the highest standards of quality and compliance.”
| Consolidated Financial Results Q3 FY2023 |
Amt in INR mn
| Particulars | Q3 FY2023 | % of sales | Q2 FY2023 | % of sales | QoQ Gr% | Q3 FY2022 | % of sales | YoY Gr% |
| Sales | 42,446 | 100.0% | 40,912 | 100.0% | 3.7% | 40,875 | 100.0% | 3.8% |
| Other operating income | 776 | 1.8% | 543 | 1.3% | 42.9% | 734 | 1.8% | 5.7% |
| Total Revenue from operations | 43,222 | 101.8% | 41,455 | 101.3% | 4.3% | 41,609 | 101.8% | 3.9% |
| Material cost | 17,071 | 40.2% | 17,128 | 41.9% | -0.3% | 16,946 | 41.5% | 0.7% |
| Gross Profit (Excl. Other op. income) | 25,375 | 59.8% | 23,784 | 58.1% | 6.7% | 23,929 | 58.5% | 6.0% |
| Employee cost | 7,640 | 18.0% | 7,716 | 18.9% | -1.0% | 7,438 | 18.2% | 2.7% |
| Manufacturing & Other expenses | 13,351 | 31.5% | 12,268 | 30.0% | 8.8% | 13,518 | 33.1% | -1.2% |
| Other Income | 180 | 0.4% | 149 | 0.4% | 20.8% | 341 | 0.8% | -47.2% |
| Forex Loss / (Gain) | (166) | -0.4% | (188) | -0.5% | -11.7% | 9 | 0.0% | – |
| EBITDA | 5,506 | 13.0% | 4,680 | 11.4% | 17.6% | 4,039 | 9.9% | 36.3% |
| Depreciation, Amortization | 2,204 | 5.2% | 2,035 | 5.0% | 8.3% | 2,034 | 5.0% | 8.4% |
| EBIT | 3,302 | 7.8% | 2,645 | 6.5% | 24.8% | 2,005 | 4.9% | 64.7% |
| Finance cost | 841 | 2.0% | 549 | 1.3% | 53.2% | 334 | 0.8% | 151.8% |
| Adjusted Profit Before Tax (PBT) | 2,461 | 5.8% | 2,096 | 5.1% | 17.4% | 1,671 | 4.1% | 47.3% |
| Business Compensation Expense | – | – | – | – | – | – | – | |
| Impairment Expense | – | – | – | – | – | – | – | |
| Profit Before Tax (PBT) | 2,461 | 5.8% | 2,096 | 5.1% | 17.4% | 1,671 | 4.1% | 47.3% |
| Tax | 885 | 2.1% | 751 | 1.8% | 17.8% | (3,820) | -9.3% | 123.2% |
| Profit After Tax (PAT) | 1,576 | 3.7% | 1,345 | 3.3% | 17.2% | 5,491 | 13.4% | -71.3% |
| (+) Share of Profit from JV | – | – | – | – | – | – | – | – |
| (-) Non-Controlling Interest | 41 | 0.1% | 48 | 0.1% | -14.6% | 36 | 0.1% | 13.9% |
| Profit/(Loss) for the period | 1,535 | 3.6% | 1,297 | 3.2% | 18.3% | 5,455 | 13.3% | -71.9% |
| | | | | | | | |
| Consolidated Financial Results – 9M FY2023 |
Amt in INR mn
| Particulars | 9M FY2023 | % of sales | 9M FY2022 | % of sales | YoY Gr% |
| Sales | 119,397 | 100.0% | 123,283 | 100.0% | -3.2% |
| Other operating income | 2,719 | 2.3% | 1,942 | 1.6% | 40.0% |
| Total Revenue from operations | 122,116 | 102.3% | 125,225 | 101.6% | -2.5% |
| Material cost | 50,296 | 42.1% | 48,490 | 39.3% | 3.7% |
| Gross Profit (Excl. Other op. income) | 69,101 | 57.9% | 74,793 | 60.7% | -7.6% |
| Employee cost | 23,142 | 19.4% | 22,862 | 18.5% | 1.2% |
| Manufacturing & Other expenses | 37,512 | 31.4% | 35,174 | 28.5% | 6.7% |
| Other Income | 361 | 0.3% | 1,269 | 1.0% | -71.6% |
| Forex Loss / (Gain) | (1,038) | -0.9% | (282) | -0.2% | 268.1% |
| EBITDA | 12,565 | 10.5% | 20,250 | 16.4% | -38.0% |
| Depreciation, Amortization | 6,167 | 5.2% | 6,238 | 5.1% | -1.1% |
| EBIT | 6,398 | 5.4% | 14,012 | 11.4% | -54.3% |
| Finance cost | 1,818 | 1.5% | 1,013 | 0.8% | 79.5% |
| Adjusted Profit Before Tax (PBT) | 4,580 | 3.8% | 12,999 | 10.5% | -64.8% |
| Business Compensation Expense1 | – | – | 18,796 | 15.2% | |
| Impairment Expense2 | – | – | 7,077 | 5.7% | |
| Profit Before Tax (PBT) | 4,580 | 3.8% | (12,874) | -10.4% | 135.6% |
| Tax | 2,527 | 2.1% | (2,896) | -2.3% | 187.3% |
| Profit After Tax (PAT) | 2,053 | 1.7% | (9,978) | -8.1% | 120.6% |
| (+) Share of Profit from JV | 0 | 0.0% | 2 | 0.0% | |
| (-) Non-Controlling Interest | 112 | 0.1% | 125 | 0.1% | -10.4% |
| Profit/(Loss) for the period | 1,941 | 1.6% | (10,101) | -8.2% | 119.2% |
| | | | | |
FY2022 includes –
- Glumetza litigation and settlement expense in Q2 FY2022
- Impairment Expense of INR 7,077 mn for impairment of Solosec® IP
Amt in INR mn
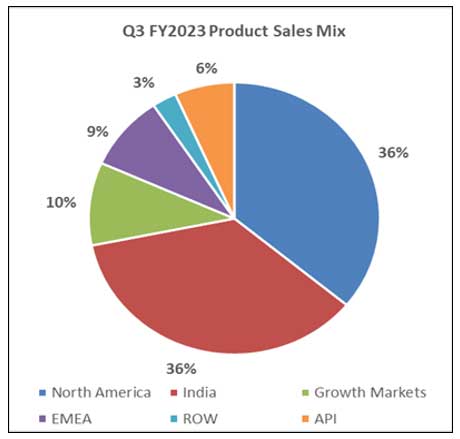
| Particulars | Q3 FY2023 | Q2 FY2023 | Growth QoQ | Q3FY2022 | Growth YoY |
| North America | 15,271 | 13,295 | 14.9% | 15,775 | -3.2% |
| India | 15,213 | 15,841 | -4.0% | 14,733 | 3.3% |
| Growth Markets | 4,187 | 4,449 | -5.9% | 3,390 | 23.5% |
| EMEA | 3,802 | 3,842 | -1.0% | 3,422 | 11.1% |
| ROW | 1,158 | 986 | 17.4% | 991 | 16.9% |
| Total Formulations | 39,631 | 38,413 | 3.2% | 38,311 | 3.4% |
| API | 2,815 | 2,499 | 12.7% | 2,564 | 9.8% |
| Consolidated Sales | 42,446 | 40,912 | 3.7% | 40,875 | 3.8% |
 North America
North America
North America sales for Q3 FY2023 were INR 15,271 mn, up 14.9% compared to INR 13,295 mn in Q2 FY2023; down 3.2% compared to INR 15,775 mn in Q3 FY2022; accounting for 36% of Lupin’s global sales.
Q3 FY2023 sales were USD 177 mn compared to USD 159 mn in Q2 FY2023 and USD 202 mn in Q3 FY2022.
The Company filed 7 ANDAs in the quarter, received 2 ANDA approvals from the U.S. FDA, and launched 4 products in the quarter in the U.S. The Company now has 166 generic products in the U.S.
Lupin continues to be the 3rd largest pharmaceutical player in both U.S. generic market and U.S. total market by prescriptions. (IQVIA Dec 2022) Lupin is the leader in 42 of its marketed generics in the U.S. and amongst the Top 3 in 107 of its marketed products (IQVIA Dec 2022).
India
India formulation sales for Q3 FY2023 were INR 15,213 mn, down 4.0% compared to INR 15,841 mn in Q2 FY2023; up 3.3% compared to INR 14,733 mn in Q3 FY2022; accounting for 36% of Lupin’s global sales.
India Region Formulations sales declined by 3.4% in the quarter as compared to Q2 FY2023; up 2.6% as compared to Q3 FY2022. The company launched 8 brands across therapies during the quarter.
Lupin is the 6th largest company in the Indian Pharmaceutical Market (IQVIA MAT Dec 2022).
Growth Markets (LATAM and APAC)
Growth Markets registered sales of INR 4,187 mn for Q3 FY2023, down 5.9% compared to INR 4,449 mn in Q2 FY2023; up 23.5% compared to INR 3,390 mn in Q3 FY2022; accounting for 10% of Lupin’s global sales.
Brazil sales were BRL 70 mn for Q3 FY2023, compared to BRL 69 mn for Q2 FY2023 and BRL 49 mn for Q3 FY2022.
Mexico sales were MXN 217 mn for Q3 FY2023, compared to MXN 224 mn for Q2 FY2023 and MXN 195 mn for Q3 FY2022.
Philippines sales were PHP 452 mn for Q3 FY2023, compared to PHP 467 mn for Q2 FY2023 and PHP 401 mn for Q3 FY2022.
Australia sales were AUD 21.1 mn for Q3 FY2023, compared to AUD 25.6 mn for Q2 FY2023 and AUD 17.8 mn for Q3 FY2022.
Europe, Middle East and Africa (EMEA)
EMEA sales for Q3 FY2023 were INR 3,802 mn, down 1.0% compared to INR 3,842 mn in Q2 FY2023; up 11.1% compared to INR 3,422 mn in Q3 FY2022; accounting for 9% of Lupin’s global sales.
South Africa sales were ZAR 308 mn for Q3 FY2023, compared to ZAR 358 mn for Q2 FY2023 and ZAR 319 mn for Q3 FY2022. Lupin is the 8th largest player in South Africa in the total generics market (IQVIA Nov 2022).
Germany sales were EUR 11.4 mn for Q3 FY2023, compared to EUR 9.9 mn for Q2 FY2023 and EUR 8.8 mn for Q3 FY2022.
Global API
Global API Sales for Q3 FY2023 were INR 2,815 mn, up 12.7% compared to INR 2,499 mn in Q2 FY2023; up 9.8% compared to INR 2,564 mn in Q3 FY2022; accounting for 6% of Lupin’s global sales.
Research and Development
Investment in R&D was INR 2,896 mn (6.8% of sales) for Q3 FY2023, compared to INR 3,546 mn (8.7% of sales) for Q3 FY2022.
Lupin received approval for 2 ANDAs from the U.S. FDA in the quarter. Cumulative ANDA filings with the U.S. FDA stand at 469 as of Dec 31, 2022, with the company having received 302 approvals to date.
The Company now has 55 First-to-File (FTF) filings including 21 exclusive FTF opportunities. Cumulative U.S. DMF filings stand at 171 as of Dec 31, 2022.
About Lupin
Lupin is an innovation-led transnational pharmaceutical company headquartered in Mumbai, India. The Company develops and commercializes a wide range of branded and generic formulations, biotechnology products, and APIs in over 100 markets in the U.S., India, South Africa, and across the Asia Pacific (APAC), Latin America (LATAM), Europe, and Middle East regions.
The Company enjoys a leadership position in the cardiovascular, anti-diabetic, and respiratory segments and has a significant presence in the anti-infective, gastro-intestinal (GI), central nervous system (CNS), and women’s health areas. Lupin is the third-largest pharmaceutical company in the U.S. by prescriptions. The company invested 8.7% of its revenue in research and development in FY22.
Lupin has 15 manufacturing sites, 7 research centers, more than 20,000 professionals working globally, and has been consistently recognized as a ‘Great Place to Work’ in the Biotechnology & Pharmaceuticals sector.
Please visit www.lupin.com for more information.
Follow us on Twitter: https://twitter.com/LupinGlobal
LinkedIn : https://www.linkedin.com/company/lupin
Facebook: http://www.facebook.com/LupinWorld/
For further information or queries please contact –
Shweta Munjal
Vice President & Global Head – Corporate Communications
Email: shwetamunjal@lupin.com




