Mumbai, November 08, 2023: Pharma major Lupin Limited [BSE: 500257 | NSE: LUPIN] reported its financial performance for the quarter ending September 30, 2023. These unaudited results were taken on record by the Board of Directors at a meeting held today.
Financial Highlights – Consolidated IND-AS
Amt in INR Mn
| Particulars | Quarter | ||||
| Q2 FY2024 | Q1 FY2024 | QoQ Growth % | Q2 FY2023 | YoY Growth % | |
| Sales | 49,392 | 47,421 | ↑4.2% | 40,912 | ↑20.7% |
| EBITDA | 9,582 | 8,791 | ↑ 9.0% | 4,680 | ↑ 104.8% |
| EBITDA Margin (%) | 19.4% | 18.5% | ↑ 90 bps | 11.4% | ↑ 800 bps |
| PBT | 6,297 | 5,588 | ↑12.7% | 2,096 | ↑200.4% |
Income Statement highlights – Q2 FY2024
- Gross Profit was INR 32,365 mn compared to INR 31,013 mn in Q1 FY2024, with gross margin of 65.5%
- Personnel cost was 17.4% of sales at INR 8,607 mn compared to INR 8,444 mn in Q1 FY2024
- Manufacturing and other expenses were 31.4% of sales at INR 15,520 mn compared to INR 14,724 mn in Q1 FY2024
- Investment in R&D for the quarter was INR 3,764 mn (7.6% of sales)
Balance Sheet highlights
- Operating working capital was INR 56,755 mn as on September 30, 2023
- Capital Expenditure for the quarter was INR 1,345 mn
- Net Debt as on September 30, 2023 stands at INR 18,056 mn
- Net Debt-Equity as on September 30, 2023 stands at 0.14
Commenting on the results, Mr. Nilesh Gupta, Managing Director, Lupin Limited said, “We delivered growth across all our target geographies, while managing costs and achieving operating leverage, thereby driving strong topline and bottomline growth. The U.S. launch of Tiotropium DPI was the highlight of the quarter, making inhalation a substantial portion of our U.S. sales. Our India business continues to do well, with growth in our target therapies ahead of market. We are also making progress on our goal of becoming best in class in quality and compliance with both our Nagpur-1 and Mandideep-2 facilities received satisfactory inspection closures”
Consolidated Financial Results Q2 FY2024
Amt in INR mn
| Particulars | Q2 FY2024 | % of sales | Q1 FY2024 | % of sales | QoQ Gr% | Q2 FY2023 | % of sales | YoY Gr% |
| Sales | 49,392 | 100.0% | 47,421 | 100.0% | 4.2% | 40,912 | 100.0% | 20.7% |
| Other operating income | 994 | 2.0% | 720 | 1.5% | 38.1% | 543 | 1.3% | 83.1% |
| Total Revenue from operations | 50,386 | 102.0% | 48,141 | 101.5% | 4.7% | 41,455 | 101.3% | 21.5% |
| Material cost | 17,027 | 34.5% | 16,408 | 34.6% | 3.8% | 17,128 | 41.9% | -0.6% |
| Gross Profit (Excl. Other op. income) | 32,365 | 65.5% | 31,013 | 65.4% | 4.4% | 23,784 | 58.1% | 36.1% |
| Employee cost | 8,607 | 17.4% | 8,444 | 17.8% | 1.9% | 7,716 | 18.9% | 11.5% |
| Manufacturing & Other expenses | 15,520 | 31.4% | 14,724 | 31.0% | 5.4% | 12,268 | 30.0% | 26.5% |
| Other Income | 404 | 0.8% | 228 | 0.5% | 77.2% | 149 | 0.4% | 171.1% |
| Forex Loss / (Gain) | 54 | 0.1% | 2 | 0.0% | 2600.0% | (188) | -0.5% | -128.7% |
| EBITDA | 9,582 | 19.4% | 8,791 | 18.5% | 9.0% | 4,680 | 11.4% | 104.7% |
| Depreciation, Amortization & Impairment Expense | 2,479 | 5.0% | 2,346 | 4.9% | 5.7% | 2,035 | 5.0% | 21.8% |
| EBIT | 7,103 | 14.4% | 6,445 | 13.6% | 10.2% | 2,645 | 6.5% | 168.5% |
| Finance cost | 806 | 1.6% | 857 | 1.8% | -6.0% | 549 | 1.3% | 46.8% |
| Profit Before Tax (PBT) | 6,297 | 12.7% | 5,588 | 11.8% | 12.7% | 2,096 | 5.1% | 200.4% |
| Tax | 1,344 | 2.7% | 1,055 | 2.2% | 27.4% | 751 | 1.8% | 79.0% |
| Profit After Tax (PAT) | 4,953 | 10.0% | 4,533 | 9.6% | 9.3% | 1,345 | 3.3% | 268.3% |
| (+) Share of Profit from JV | – | – | – | – | – | – | – | |
| (-) Non-Controlling Interest | 56 | 0.1% | 10 | 0.0% | 460.0% | 48 | 0.1% | 16.7% |
| Profit/(Loss) for the period | 4,897 | 9.9% | 4,523 | 9.5% | 8.3% | 1,297 | 3.2% | 277.6% |
Consolidated Financial Results H1 FY2024
Amt in INR mn
| Particulars | H1 FY2024 | % of sales | H1 FY2023 | % of sales | YOY Gr% |
| Sales | 96,813 | 100.0% | 76,951 | 100.0% | 25.8% |
| Other operating income | 1,713 | 1.8% | 1,943 | 2.5% | -11.8% |
| Total Revenue from operations | 98,526 | 101.8% | 78,894 | 102.5% | 24.9% |
| Material cost | 33,435 | 34.5% | 33,226 | 43.2% | 0.6% |
| Gross Profit (Excl. Other op. income) | 63,378 | 65.5% | 43,725 | 56.8% | 44.9% |
| Employee cost | 17,051 | 17.6% | 15,501 | 20.1% | 10.0% |
| Manufacturing & Other expenses | 30,233 | 31.2% | 24,185 | 31.4% | 25.0% |
| Other Income | 622 | 0.6% | 205 | 0.3% | 203.4% |
| Forex Loss / (Gain) | 56 | 0.1% | (872) | -1.1% | -106.4% |
| EBITDA | 18,373 | 19.0% | 7,059 | 9.2% | 160.3% |
| Depreciation, Amortization & Impairment Expense | 4,825 | 5.0% | 3,963 | 5.2% | 21.8% |
| EBIT | 13,548 | 14.0% | 3,096 | 4.0% | 337.6% |
| Finance cost | 1,663 | 1.7% | 977 | 1.3% | 70.2% |
| Profit Before Tax (PBT) | 11,885 | 12.3% | 2,119 | 2.8% | 461.0% |
| Tax | 2,399 | 2.5% | 1,642 | 2.1% | 46.1% |
| Profit After Tax (PAT) | 9,486 | 9.8% | 477 | 0.6% | 1888.7% |
| (+) Share of Profit from JV | – | – | – | – | – |
| (-) Non-Controlling Interest | 67 | 0.1% | 70 | 0.1% | -4.3% |
| Profit/(Loss) for the period | 9,419 | 9.7% | 407 | 0.5% | 2214.3% |
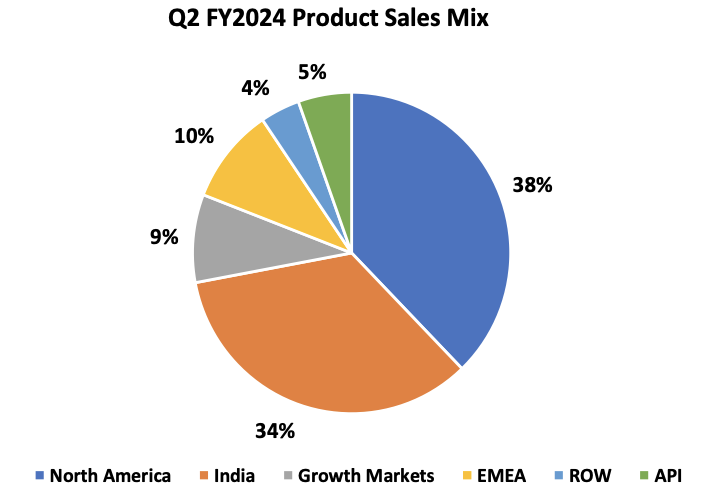
Sales Mix
Amt in INR mn
| Particulars | Q2 FY2024 | Q1 FY2024 | Growth QoQ | Q2 FY2023 | Growth YoY |
| North America | 18,666 | 15,905 | 17.4% | 13,295 | 40.4% |
| India | 16,915 | 16,384 | 3.2% | 15,841 | 6.8% |
| Growth Markets | 4,378 | 4,066 | 7.7% | 4,449 | -1.6% |
| EMEA | 4,759 | 3,987 | 19.4% | 3,842 | 23.9% |
| ROW | 1,990 | 1,655 | 20.2% | 986 | 101.8% |
| Total Formulations | 46,708 | 41,997 | 11.2% | 38,413 | 21.6% |
| API | 2,684 | 3,371 | -20.4% | 2,499 | 7.4% |
| Total Product Sales | 49,392 | 45,368 | 8.9% | 40,912 | 20.7% |
| NCE Income | – | 2053 | NA | – | NA |
| Consolidated Sales | 49,392 | 47,421 | 4.2% | 40,912 | 20.7% |
Operational Highlights
North America
North America sales for Q2 FY2024 were INR 18,666 mn, up 17.4% compared to INR 15,905 mn in Q1 FY2024; up 40.4% as compared to INR 13,295 mn in Q2 FY2023; and accounted for 38% of Lupin’s global sales.
Q2 FY2024 sales were USD 213 mn compared to USD 181 mn in Q1 FY2024 and USD 159 mn in Q2 FY2023.
The Company received 11 ANDA approvals from the U.S. FDA, and launched 1 product in the quarter in the U.S. The Company now has 157 generic products in the U.S.
Lupin continues to be the 3rd largest pharmaceutical player in both U.S. generic market and U.S. total market by prescriptions (IQVIASeptember 2023). Lupin is the leader in 48 of its marketed generics in the U.S. and amongst the Top 3 in 101 of its marketed products (IQVIA September 2023).
India
India sales for Q2 FY2024 were INR 16,915 mn, up 3.2% as compared to INR 16,384 mn in Q1 FY2024; up 6.8% as compared to INR 15,841 mn in Q2 FY2023; and accounted for 34% of Lupin’s global sales.
India Region Formulations sales grew by 2.6% in the quarter as compared to Q1 FY2024, up 6.9% as compared to Q2 FY2023. The company launched 4 brands across therapies during the quarter.
Lupin is the 7th largest company in the Indian Pharmaceutical Market (IQVIA MAT September 2023).
Growth Markets (LATAM and APAC)
Growth Markets registered sales of INR 4,378 mn for Q2 FY2024, growing 7.7% compared to INR 4,066 mn in Q1 FY2024; down 1.6% as compared to INR 4,449 mn in Q2 FY2023; and accounted for 9% of Lupin’s global sales.
Brazil sales were BRL 74 mn for Q2 FY2024, compared to BRL 75 mn for Q1 FY2024 and BRL 69 mn for Q2 FY2023.
Mexico sales were MXN 83 mn for Q2 FY2024, compared to MXN 86 mn for Q1 FY2024 and MXN 224 mn for Q2 FY2023.
Philippines sales were PHP 621 mn for Q2 FY2024, compared to PHP 463 mn for Q1 FY2024 and PHP 467 mn for Q2 FY2023.
Australia sales were AUD 26 mn for Q2 FY2024, compared to AUD 26 mn for Q1 FY2024 and AUD 26 mn for Q2 FY2023.
Europe, Middle-East and Africa (EMEA)
EMEA sales for Q2 FY2024 were INR 4,759 mn, up 19.4% compared to INR 3,987 mn in Q1 FY2024; up 23.9% compared to INR 3,842 mn in Q2 FY2023; and accounted for 10% of Lupin’s global sales.
South Africa sales were ZAR 341 mn for Q2 FY2024, compared to ZAR 295 mn for Q1 FY2024 and ZAR 358 mn for Q2 FY2023. Lupin is the 7th largest player in South Africa in the total generics market (IQVIA September 2023).
Germany sales were EUR 13 mn for Q2 FY2024, compared to EUR 10 mn for Q1 FY2024 and EUR 10 mn for Q2 FY2023.
Global API
Global API Sales for Q2 FY2024 were INR 2,684 mn, down 20.4% as compared to INR 3,371 mn in Q1 FY2024; up 7.4% as compared to INR 2,499 mn in Q2 FY2023; and accounted for 5% of Lupin’s global sales.
Research and Development
Investment in R&D was INR 3,764 mn (7.6% of sales) for Q2 FY2024 as compared to INR 3,376 mn (8.3% of sales) for Q2 FY2023.
Lupin received approval for 11 ANDAs from the U.S. FDA in the quarter. Cumulative ANDA filings with the U.S. FDA stand at 444 as of September 30, 2023, with the company having received 305 approvals to date.
The Company now has 53 First-to-File (FTF) filings including 20 exclusive FTF opportunities. Cumulative U.S. DMF filings stand at 167 as of September 30, 2023.
About Lupin
Lupin is an innovation-led transnational pharmaceutical company headquartered in Mumbai, India. The Company develops and commercializes a wide range of branded and generic formulations, biotechnology products, and APIs in over 100 markets in the U.S., India, South Africa, and across the Asia Pacific (APAC), Latin America (LATAM), Europe, and Middle East regions.
The Company enjoys a leadership position in the cardiovascular, anti-diabetic, and respiratory segments and has a significant presence in the anti-infective, gastro-intestinal (GI), central nervous system (CNS), and women’s health areas. Lupin is the third-largest pharmaceutical company in the U.S. by prescriptions. The company invested 7.9% of its revenue in research and development in FY23.
Lupin has 15 manufacturing sites, 7 research centers, more than 20,000 professionals working globally, and has been consistently recognized as a ‘Great Place to Work’ in the Biotechnology & Pharmaceuticals sector.
Please visit www.lupin.com for more information.
Follow us on:
LinkedIn: https://www.linkedin.com/company/lupin
Facebook: http://www.facebook.com/LupinWorld/
Twitter: https://twitter.com/LupinGlobal
For further information or queries please contact –
Shweta Munjal
Vice President & Global Head – Corporate Communications & Sustainability
Email: shwetamunjal@lupin.com